The thyroid gland, a tiny organ shaped like a butterfly and found in the neck, is incredibly important for controlling the body’s metabolism. Its main job is to create thyroid hormones, which are absolutely essential for keeping our bodies functioning properly.
In this post, we’re going to discuss thyroid hormone production, uncovering the intricate processes that make it all happen.
Overview
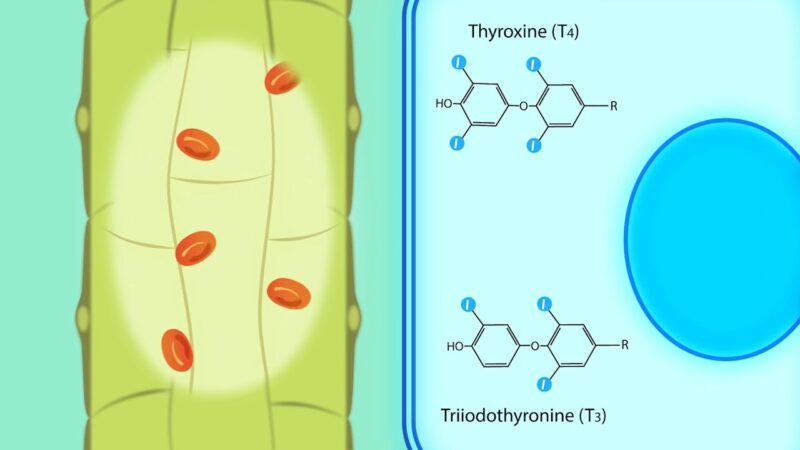
Thyroid Hormones are special hormones that belong to a group called amines, and they are made using an important building block called tyrosine, which is an amino acid.
The thyroid gland, which is the main factory for these hormones, produces a whopping twenty times more of a hormone called T4 compared to another hormone called T3.
Later on, T4 can be transformed into either T3 or rT3 with the help of an enzyme called ‘Iodinase’ that can be found in various tissues throughout the body.
Functional Thyroid Gland Histology
The thyroid gland is packed with thyroid follicles, which are the building blocks responsible for making thyroid hormones. These follicles are surrounded by a layer of follicular epithelial cells and house a protein-filled space called the Thyroid Colloid.
The production of thyroid hormones is a sophisticated process that takes place within the follicular epithelial cells and the protein-filled space of the follicles.
Thyroid Hormone Synthesis Mechanism
Iodine Transport
Iodine plays an important role in producing thyroid hormones.
The special thing is that iodine takes the form of iodide (I-) when it’s in its ionic state, and it gets transported from our bloodstream into the follicular lumen by these amazing cells called Follicular Epithelial Cells.
This process leads to a high concentration of iodide specifically in the thyroid gland, way more than in the rest of our body.
The iodide cycle consists of a series of transport, oxidation and coupling steps in thyroid follicular cells to produce thyroid hormone. – Angela Leung
Thyroglobulin Synthesis
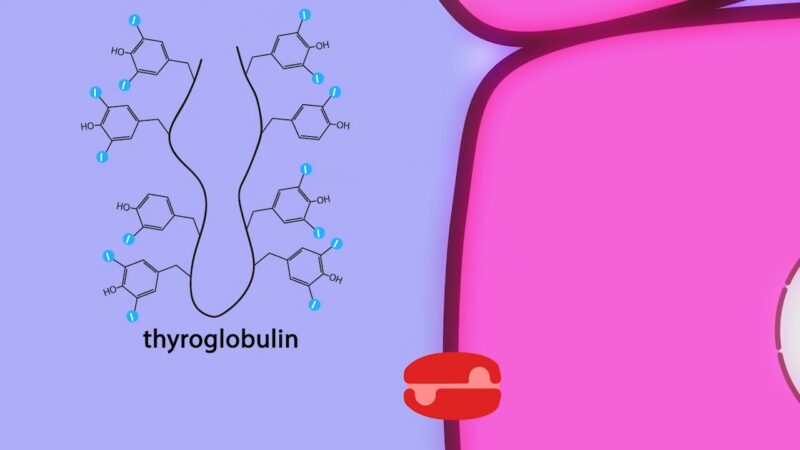
Thyroglobulin, a protein filled with numerous tyrosine amino acids, transforms into distinct thyroid hormone molecules. This incredible process takes place inside the follicular epithelial cell, where thyroglobulin is carefully synthesized and then released into the follicular lumen.
Thyroglobulin (Tg) is a vertebrate secretory protein synthesized in the thyrocyte endoplasmic reticulum (ER), where it acquires N-linked glycosylation and conformational maturation (including formation of many disulfide bonds), leading to homodimerization. – Bruno Di Jeso
Thyroid Peroxidase
Thyroid Peroxidase is an enzyme present in the acellular colloid of the follicular lumen, performing several key reactions:
Notably, the thyroid gland primarily produces T4 rather than T3 due to the efficiency of combining two DIT residues.
Endocytosis of Peroxidase-processed Thyroglobulin
Peroxidase-processed thyroglobulin is endocytosed by follicular epithelial cells when the thyroid gland is stimulated to release thyroid hormone into circulation. This reservoir of it can last for months, explaining why defects in thyroid hormone synthesis often take months to become clinically apparent.
Release of T4 and T3 from Thyroglobulin
Once endocytosed into the follicular epithelial cell, thyroglobulin is broken down by lysosomes, releasing attached T4, T3, MIT, and DIT.
T4 and T3 are then transported into the circulation, while the iodine atoms of MIT and DIT are salvaged and transported back into the follicular lumen as I-.